Stem cells are among the most important biological tools in modern medicine because they can renew themselves and, under the right conditions, develop into more specialized cell types. That combination makes them central to regenerative medicine, disease modelling and future therapeutic strategies.
Their relevance is not only scientific. Stem cells are already used in some established treatments, and they are also part of some of the most promising areas of medical research, from tissue repair to more personalized therapeutic approaches.
Stem cells matter because they connect basic biology with some of the most advanced goals in medicine: regeneration, repair, modeling of disease and next-generation therapies.
What are stem cells?
A stem cell is a type of cell with two defining properties. First, it can divide and produce more cells like itself. Second, it can differentiate into other, more specialized cell types depending on the signals and environment around it.
These properties make stem cells valuable both in natural biology and in medicine. They are involved in development, tissue renewal and repair, and they also offer powerful possibilities for laboratory research and therapeutic innovation.

Stem cells are not important just because they can grow, but because they can become something else under the right conditions.
Main types of stem cells
Stem cells are usually classified by origin and biological potential. Each category has different medical value, technical challenges and ethical implications.
Embryonic and pluripotent routes
These stem cells have the broadest differentiation potential, which makes them scientifically powerful but also ethically more sensitive depending on the source.
Adult, cord and related routes
These are often more restricted in what they can become, but they remain highly relevant in established treatments and regenerative medicine research.
The best type of stem cell depends on the medical objective, not only on biological potential in abstract terms.
Four powerful uses of stem cells in modern medicine
Stem cells are not one single therapy. Their value appears across multiple medical and scientific applications, some already established and others still advancing quickly.
Stem cells are already used in clinical practice, especially in bone marrow and related transplant strategies for diseases such as leukemia and lymphoma.
They are being studied to help repair or replace damaged tissues in organs such as the heart, liver, nervous system and other structures.
Stem cells help researchers recreate disease conditions in the lab and test treatments in more biologically relevant systems.
They are part of the broader movement toward more personalized and engineered therapeutic strategies in advanced medicine.
Main challenges in stem cell medicine
Despite their potential, stem cells also bring real challenges. The field has to deal with ethical debates, biological risks, process control issues and the fact that many promising therapies are still expensive and difficult to scale.
Stem cells are full of promise, but the real challenge is turning biological potential into safe, controlled and scalable medicine.
Why stem cells matter for bioprocessing
Stem cell medicine does not depend only on biology. It also depends on process technology. If stem cells are going to move from laboratory promise toward broader therapeutic reality, they need controlled handling, reproducible cultivation environments and scalable workflows.
This is where bioprocessing becomes especially relevant. The future of stem cell applications is linked not only to differentiation and clinical insight, but also to automation, contamination control, scale strategy and high-quality consumables.

How TECNIC fits this workflow
TECNIC fits naturally into this topic through the connection between advanced cell processes and controlled bioprocess systems. Stem cell workflows increasingly depend on reliable cultivation, controlled environments and scalable process routes, which is where bioreactors, single-use components and related solutions become relevant.
Bioreactors
Relevant when stem cell processes need controlled cultivation conditions from laboratory development to larger-scale process work.
Single-use solutions
Useful where sterility, flexibility and lower cross-contamination risk are especially important in advanced cell workflows.
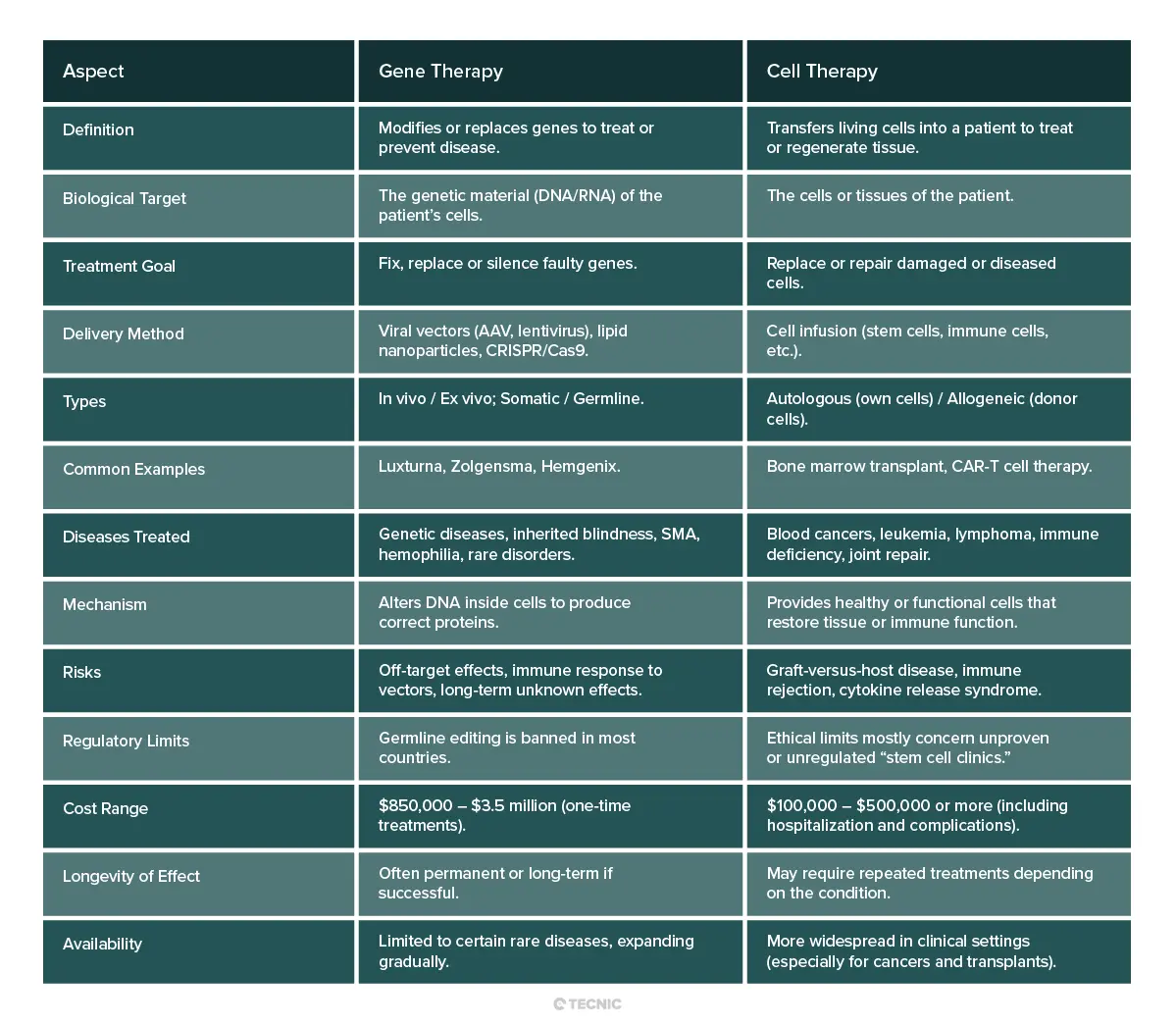
Cell and gene therapy context
Stem cells connect naturally with the broader advanced therapy area already reflected in TECNIC content.
Contact TECNIC
When stem cell applications turn into real process questions, direct technical discussion becomes far more useful than a generic overview.
This article works best when stem cells are presented as both a medical topic and a process challenge, because that is where TECNIC’s role becomes meaningful.
Frequently asked questions
What are stem cells?
Stem cells are cells that can renew themselves and also develop into more specialized cell types under the right conditions.
Why are stem cells important in medicine?
Because they are relevant for regenerative medicine, treatment of some diseases, disease modelling and future advanced therapies.
What are the main types of stem cells?
Broadly, they include embryonic, adult, umbilical cord and induced pluripotent stem cells.
What are stem cells used for today?
They are already used in some blood-related treatments and are also studied for tissue repair, drug testing and future cell-based therapies.
What is the biggest challenge in stem cell medicine?
One of the biggest challenges is turning biological potential into safe, reproducible and scalable therapies.
Exploring how advanced cell processes connect with scalable bioprocessing?
Explore TECNIC’s bioprocess solutions or speak with our team to review the right path for controlled cell-based workflows.